Formations of Ionic Bonds and Properties:
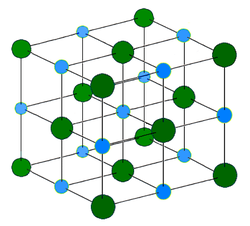
An ionic coumpound is made up of positive ions (cations) and negative ions (anions) held together be electrostatic forces in a rigid array or lattice. Ionic bonding refers to the electrostatic attract between cations and anions.
The physical properties of ionic compounds are:
- A. High melting and boiling point.
- B. Ionic solids do not conduct electricity (they are insulators).
- C. When molten (liquid) ionic compounds conduct electricity.
- D. When dissolved in water to form an aqueous solution ionic compounds conduct electricity.
- E. Hard
- F. Brittle
