What is an Ionic Compound?
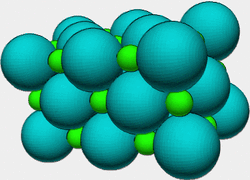
An ionic compound is a chemical compound in which ions are held together in a lattice structure by ionic bonds. Usually, the positively charged portion consists of cations and the negatively charged portion is an anion or polyatomic ion. Ions in ionic compounds are held together by the electrostatic force between oppositely charged bodies. Basically it is a bond between a positively charged molecule (cation) and a negatively charged molecule (anion). Table Salt is a good Ionic Compound, because the Na is positive and the Cl is negative.
Examples...
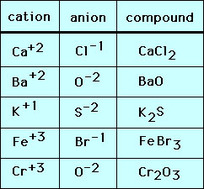
Sodium Chloride- NaCl Barium Chloride- BaCl2
Magnesium Fluoride- MgF2 Magnesium Nitride- Mg3N
Caesium Chloride- CsCl Sodium Sulphide- Na2S
Magnesium Fluoride- MgF2 Magnesium Nitride- Mg3N
Caesium Chloride- CsCl Sodium Sulphide- Na2S

